What Is Malignant Epithelioid Mesothelioma?
Epithelioid mesothelioma is a form of malignant mesothelioma made up of epithelioid cells. It is a form of cancer caused by asbestos.
Epithelial cells make up one of four types of tissues naturally found in the body. Epithelial tissue is located on the skin’s surface, in the linings of hollow organs and blood vessels and along the lining of internal cavities.
When an individual is exposed to asbestos fibers, epithelial cells can mutate and become cancerous. Epithelioid mesothelioma is the most common form of the disease. The cell type is found in about 50% – 70% of all mesothelioma cancer patients.
Epithelioid Mesothelioma Key Facts
Some important facts about epithelioid mesothelioma include:
- It is the most common mesothelioma cell type.
- 50% to 70% of all mesothelioma cases are epithelioid.
- It responds well to treatment compared to other cell types.
- This type commonly qualifies for multimodal treatment.
- With treatment, survival ranges from 18 months to more than 3 years.
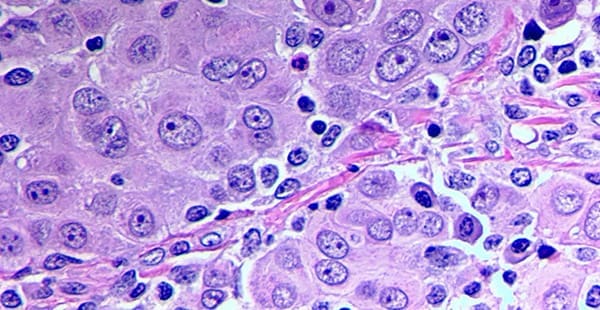
- Epithelioid cells can be a variety of shapes, including cubed, columnar and squamous (flat).
Squamous cells are commonly found in the lining of the lungs, also known as the pleura. Epithelioid mesothelioma cells are also recognizable compared to other mesothelioma cell types because of how they adhere to each other. The cells form in small clusters and may be round or oval. The cells can also be identified by their visible nuclei.
Epithelioid cells typically divide faster than other cells. However, the way the cells form in clusters may make them slower to metastasize compared to other cell types.
Resources for Mesothelioma Patients
Epithelial Mesothelioma Symptoms
Malignant epithelioid mesothelioma can present with a variety of symptoms, depending on where the cancer develops. The presence and severity of symptoms also depend on a patient’s overall health, age, the cell type and the stage at diagnosis.
Epithelioid mesothelioma patients may not experience severe symptoms until a later stage of disease. Fever and fluid buildup around the lungs or in the abdomen are common symptoms of epithelioid mesothelioma.
Epithelial Mesothelioma Diagnosis
The diagnostic process for epithelioid mesothelioma typically begins with a series of imaging tests. These tests may include X-rays, CT scans, PET scans or MRIs. If a tumor or abnormality is detected, a blood test may be ordered. Blood tests can identify specific biomarkers to help determine if mesothelioma cells are present.
A biopsy will be performed to confirm a diagnosis of epithelioid mesothelioma. A biopsy can be a non-surgical or surgical procedure to remove a tissue sample for analysis. Lab tests can determine the type of cells present. If they have the right characteristics, these tests can confirm a diagnosis of epithelioid mesothelioma.
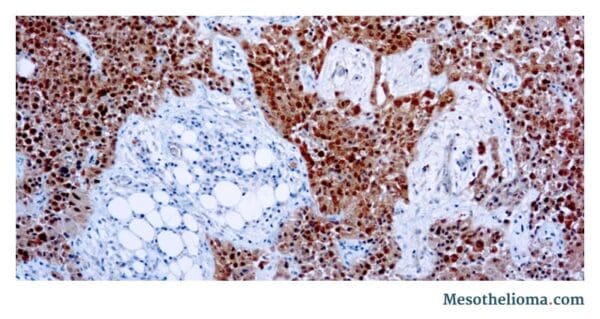
Immunohistochemistry for Diagnosing Epithelioid Mesothelioma
Guidelines recommend immunohistochemistry (IHC) testing as a way to confirm an epithelioid mesothelioma diagnosis. It is a staining technique that can identify cell type and differentiate mesothelioma from other cancers, such as adenocarcinoma.
Sarcomatoid cells may also be present in these samples, which can indicate biphasic mesothelioma.
Certain antibodies can help identify epithelioid mesothelioma in patients. When these antibodies are applied in combination, they can help a pathologist accurately diagnose the disease.
Immunohistochemical Markers Used in Diagnosing Epithelioid Mesothelioma
The presence of epithelioid mesothelioma markers supports a diagnosis of epithelioid mesothelioma. Markers of epithelioid mesothelioma include:
- Calretinin: Present in almost all epithelioid mesotheliomas
- Cytokeratin: Present in 70% to 95% of mesotheliomas
- Podoplanin (D2-40): Present in 90% to 100% of mesotheliomas
Source: Archives of Pathology & Laboratory Medicine
Lung adenocarcinoma markers may help rule out a diagnosis of epithelioid mesothelioma. Markers of lung adenocarcinoma include:
- Claudin 4: Present in almost all lung adenocarcinomas and absent in mesotheliomas
- CEA: Present in 80% to 100% of lung adenocarcinomas
- MOC31: Present in 95% to 100% of lung adenocarcinomas
Rare Types of Epithelial Mesothelioma Cells
In addition to the common epithelioid cell type, pathologists have identified various rare subtypes that develop in some patients. These subtypes may have limited treatment options compared to standard epithelial mesothelioma.
- Adenomatoid mesothelioma: Adenomatoid cells, also known as glandular or microglandular mesothelioma, form in glandular structures. The cells are commonly flat or cube-shaped and can be benign or malignant.
- Adenoid cystic mesothelioma: This rare form of mesothelioma is most commonly found in the peritoneal or pleural cavities. Cystic cells are often benign, though malignancies can sometimes form. Cystic mesothelioma is more commonly diagnosed in young women, particularly those of child-bearing age.
- Deciduoid mesothelioma: This rare subtype of mesothelioma typically develops in the abdomen, but it can also form in the lining of the lungs. The cancer’s exact cause is currently unknown, and it is most often diagnosed in women.
- Small cell mesothelioma: This subtype is sometimes mistaken for small cell lung cancer. It forms in the peritoneum and pleura. Small cell mesothelioma is usually diagnosed in biphasic tumors, which contain epithelial and sarcomatoid cells.
- Well-differentiated papillary mesothelioma (WDPM): This type of mesothelioma usually forms in the pleura, peritoneum and genitals. Papillary mesothelioma is slow to metastasize. It is most often seen in young women but may develop in men. Studies have found survival times ranging from 36 to 180 months.
Epithelioid Mesothelioma Prognosis
A patient’s cell type also plays a role in prognosis. Patients with epithelioid cancer cells have a more favorable prognosis and overall life expectancy than those with other cell types. In general, patients with epithelioid mesothelioma survive 1.5 – 6.5 years.
One 2017 study examined the survival of pleural mesothelioma patients with the epithelioid cell type. The study also included patients with sarcomatoid and biphasic cell types. Researchers found the following median survivals for patients treated with multimodal therapy:
- Epithelial mesothelioma: ~23 months
- Biphasic mesothelioma: ~14 months
- Sarcomatoid mesothelioma: ~11 months
The prognosis for epithelioid mesothelioma patients depends on a number of factors, including the location and staging of mesothelioma, as well as the patient’s age, general health and pre-existing conditions.
Epithelioid Mesothelioma Treatment
Epithelioid mesothelioma treatment may include surgery, chemotherapy and radiation therapy. In many cases, patients will undergo a multimodal treatment plan, which is when two or more treatment methods are combined. Some procedures can also be used as palliative treatments, which can lessen symptoms for patients.
| Pleural | Peritoneal | |
|---|---|---|
| Checkpoint Inhibitor Immunotherapy | ~19 months | More than 39 months* |
| Chemotherapy | ~17 months | Not reported |
| Surgery + Heated Chemotherapy | ~42 months | 79 months |
*Patients received a checkpoint inhibitor after systemic chemotherapy
Sources: The Lancet, Journal of Clinical Oncology, Journal of Thoracic Oncology, Cancer
Surgery for Epithelioid Mesothelioma
Epithelial pleural mesothelioma patients may be eligible for surgery, including pleurectomy/decortication (P/D) and extrapleural pneumonectomy (EPP). These surgeries involve removing cancerous tissue from the lung and surrounding areas.
A 2018 study treated pleural mesothelioma patients with surgery and heated chemotherapy. Patients with epithelial tumors survived 42 months. Patients with sarcomatoid or biphasic tumors lived about 21 months.
This survival gap may be tied to differences in how each cell type responds to treatment. Epithelial cells may be more susceptible to chemotherapy than sarcomatoid cells, making the treatment more effective.
Multimodal Treatment for Epithelioid Mesothelioma
Patients commonly receive multimodal treatment, which is a combination of two or more treatments. This approach may kill mesothelioma cells more effectively by targeting them with more than one therapy. Combinations often include surgery, chemotherapy and sometimes radiation.
Multimodal treatment generally does the best job of extending patient survival. For example, patients with epithelioid mesothelioma who underwent P/D and heated chemotherapy had a median survival of 3.5 years. In contrast, patients treated with chemotherapy or immunotherapy alone generally live closer to 1.5 years.
Some patients may not qualify for surgery-based treatment at first. But surgery eligibility can change if the patient responds well to another treatment. Patients should discuss their treatment options with an experienced mesothelioma specialist. An experienced mesothelioma doctor can help patients understand their eligibility for multimodal treatments.
Immunotherapy in Epithelioid Mesothelioma
Immunotherapy treatments have become standard for epithelioid mesothelioma patients. Immunotherapy allows the immune system to fight cancer. Several treatments fall into this category, but immune checkpoint inhibitors (ICIs) are most common. The ICI drugs Opdivo® (nivolumab) and Yervoy® (ipilimumab) have approval for treating inoperable pleural mesothelioma. In a study, this drug combo extended survival better than chemotherapy.
Epithelioid patients may receive ICIs if they do not qualify for surgery. Patients with epithelioid mesothelioma may also be able to join clinical trials to find additional treatments that may extend life expectancy.
Chemotherapy for Epithelioid Mesothelioma
Chemotherapy uses chemicals to kill cancerous cells and prevent them from multiplying. It is a recommended treatment for many patients with epithelioid mesothelioma. Patients who do not qualify for surgery commonly receive chemotherapy instead. Epithelioid mesothelioma patients treated with chemotherapy alone have a life expectancy of about 17 months.
Chemotherapy is given in two basic forms: systemic and local (intracavitary). Systemic chemotherapy travels throughout the body. Patients may receive it as an infusion or a pill. It may be a solo treatment or part of a multimodal plan.
The drug Avastin® (bevacizumab) may boost the cancer-fighting power of chemotherapy. In a study, adding Avastin improved survival by 3 months compared to chemotherapy alone.
Local chemotherapy confines the drugs to a specific part of the body, like the chest or abdomen. This puts them in direct contact with the cancer cells and may help minimize side effects. Local chemotherapy generally occurs within a multimodal treatment plan.
Other Treatment Options for Epithelioid Mesothelioma
In some cases, patients may receive other treatments alone or in combination. Patients may also explore treatment with experimental therapies through clinical trials.
Radiation Therapy
Radiation therapy (radiotherapy) uses energy to kill cancer cells. It may be used alongside other treatments for epithelioid pleural mesothelioma. In one study, epithelioid patients who underwent surgery-based treatment and radiation had a median survival of about 3.5 years.
Medical literature rarely mentions radiotherapy as a treatment for peritoneal mesothelioma. Patients wondering about radiation should discuss it with an experienced mesothelioma specialist.
Emerging Epithelioid Treatment Options
There are a variety of experimental mesothelioma treatments. Some have promising early results while others have not yet been fully verified. Experimental therapies include pressurized chemotherapy (PIPAC), gene therapy and photodynamic therapy.
Experimental treatments may be an option for patients who did not respond to traditional therapy.
Common Questions About Epithelioid Mesothelioma
- Is epithelioid mesothelioma a type of malignant mesothelioma?
Epithelioid mesothelioma is a subtype of malignant mesothelioma cancer and is caused by asbestos. It is the most common of the three main mesothelioma cell types. Warning signs include chest pain and shortness of breath.
- What causes epithelial mesothelioma?
Asbestos exposure causes epithelioid mesothelioma. When a person inhales or ingests asbestos, the small fibers can get stuck in the body. Over time, they may reach the mesothelial cells, which make up the lining around organs and body cavities. The asbestos fibers irritate the mesothelial cells and cause inflammation. The irritation and inflammation can trigger processes that cause the cells to become cancerous. When this results in cancerous epithelioid cells, the cells are epithelioid mesothelioma.
- How does a mesothelioma cell type affect my prognosis and treatment?
Mesothelioma cell type affects prognosis and treatment because it helps doctors determine the best treatment approach. Epithelioid mesothelioma may respond best to multimodal treatment that includes surgery. Sarcomatoid and biphasic mesothelioma may respond best to immunotherapy.
Note: This page contains statistics gleaned from large groups of patients. These statistics cannot forecast a single patient’s risk, cancer experience or the success of any given treatment. Patients should discuss all cancer screening and treatment decisions with an experienced oncologist.