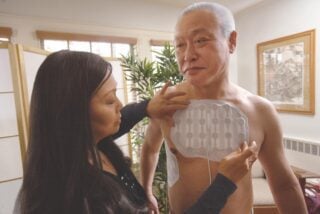
Tumor Treating Fields (TTFields) are a relatively new treatment for mesothelioma patients. TTFields uses low-intensity electrical currents to disrupt cell division. By stopping cell division, TTFields could slow or stop cancer growth.
In 2019, the U.S. Food and Drug Administration (FDA) approved the NovoTTF-100L™ System combined with pemetrexed and platinum-based chemotherapy. The treatment combination is a first-line treatment for pleural mesothelioma patients. TTFields availability has been expanding across the United States since this approval.
Who Can Receive Tumor Treating Fields for Mesothelioma?
Patients should discuss eligibility for TTFields with their physician. The treatment continues to be researched in clinical studies. The FDA approval was specifically for:
- The Novocure® NovoTTF-100L™ System, now under brand name Optune Lua™
- Patients with malignant pleural mesothelioma
- Patients with an unresectable, locally advanced or metastatic diagnosis
- Use as a first-line treatment
- Use with pemetrexed and platinum-based chemotherapy
Patients may not be eligible if they have an electronic medical device. This includes a pacemaker or defibrillator. More testing is needed to determine safety with electronic medical devices.
Where Can Mesothelioma Patients Receive TTFields Treatment?
Tumor Treating Fields for mesothelioma are available through clinical studies. Because of success in clinical research, the treatment is becoming increasingly available across the United States. However, not all cancer centers offer the treatment for mesothelioma patients.
A total of 69 cancer centers currently offer Optune Lua™. A complete list is available on the Optune Lua™ website. A few notable cancer centers offering the mesothelioma treatment include:
- Banner MD Anderson Cancer Center
- University of California Los Angeles (UCLA) Medical Center
- Moffitt Cancer Institute
- Brigham and Women’s Hospital
- Cleveland Clinic
Moffitt Cancer Center is one of the newest cancer centers that announced its plans to offer TTFields to pleural mesothelioma patients. Specialists will offer TTFields at the Tampa, Florida location.
How Can Tumor Treating Fields Help Mesothelioma Patients?
Tumor treating fields have been successful in extending survival for mesothelioma patients.
In one study, researchers focused on 80 patients with pleural mesothelioma. These patients had not received other treatment and were ineligible for surgery. Doctors treated the patients with Optune Lua™ and chemotherapy and found:
- A 1-year survival rate of 62%
- A 2-year survival rate of 42%
A follow-up CT scan was performed on 72 patients. Of these patients:
- 40% had tumors shrink
- 57% had tumors stop growing
TTFields treatment also offers convenience to mesothelioma patients. Surgery, chemotherapy and radiation therapy require hospitalization or frequent hospital visits. The Optune Lua™ system is wearable and portable. This allows patients to receive treatment from the comfort of their homes.
Patients can wear Optune Lua™ with the system’s backpack. The backpack holds the TTFields device, which is controlled by rechargeable batteries. Patients may also opt to receive treatment while they sleep.
Treatment results can vary from patient to patient. Some individuals may be better suited for other treatment options. Ultimately, a mesothelioma specialist can provide the best insight. They will help patients establish a treatment plan unique to their situation.




